 When reading the ISO 22716 standard, what draws attention, for the industrial pharmacist I am, it is the choice of the expression “it is proper to” for any given directive, where the GMPs use the expression “it is due to “. The choice of words represents one of the major differences. But what are the big differences between GMP and ISO 22716?Ĭertainly the number of pages constituting each document is already a big difference (more than 300 pages in total for the GMP, counting the Annexes and the GMP for the active substances against less than fifty for the 22716 …) but we will not go on this ground for this article. 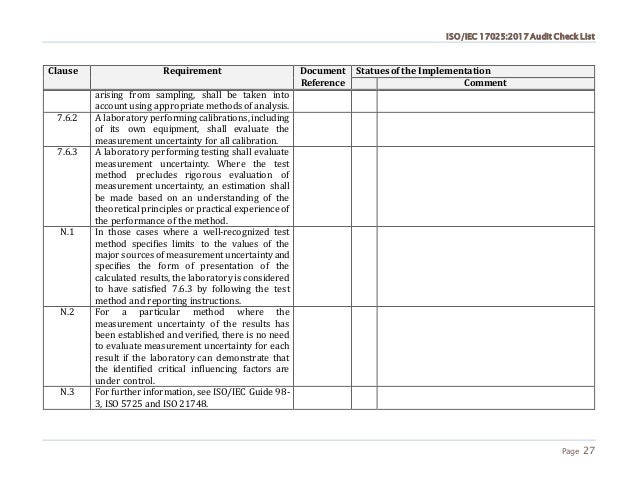
The ISO 22716 standard is intended to be THE reference for the industrials of the market. Indeed these “substances or mixtures intended to be put in contact with the superficial parts of the human body” (extract article 2, cosmetic regulation and article L.5131-1 of the CSP) can be dangerous if their production is not controlled at least. 
If the need to secure the production of drugs is not to prove, it is not always obvious for cosmetic products. 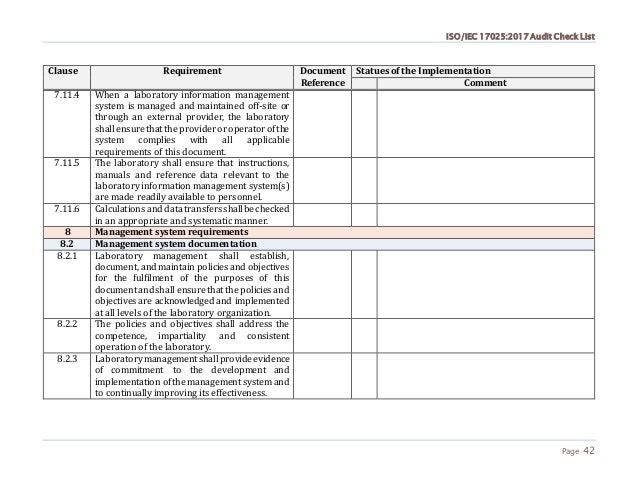
Drugs have the Good Manufacturing Practices (hereinafter referred to as GMP), cosmetics have the ISO 22716 standard. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
